[vc_row type=”in_container” full_screen_row_position=”middle” column_margin=”default” column_direction=”default” column_direction_tablet=”default” column_direction_phone=”default” scene_position=”center” text_color=”dark” text_align=”left” row_border_radius=”none” row_border_radius_applies=”bg” overlay_strength=”0.3″ gradient_direction=”left_to_right” shape_divider_position=”bottom” bg_image_animation=”none”][vc_column column_padding=”no-extra-padding” column_padding_tablet=”inherit” column_padding_phone=”inherit” column_padding_position=”all” column_element_spacing=”default” background_color_opacity=”1″ background_hover_color_opacity=”1″ column_shadow=”none” column_border_radius=”none” column_link_target=”_self” gradient_direction=”left_to_right” overlay_strength=”0.3″ width=”1/1″ tablet_width_inherit=”default” tablet_text_alignment=”default” phone_text_alignment=”default” bg_image_animation=”none” border_type=”simple” column_border_width=”none” column_border_style=”solid”][vc_column_text]
In the past couple of years, life science organizations learned how to communicate and collaborate using more virtual, tech-enabled tactics. Asynchronous virtual engagement quickly became vital to teams with a global reach, and many saw the need to rely on technology to manage input from ever-multiplying channels. Now, in-person conferences and gatherings are back. But many pharmaceutical and medical device teams are opting to maintain a virtual-first approach for reasons that center around inclusivity, resource efficiency, and flexibility. Here are some of the most compelling reasons leaders will be more intentional about virtual engagement and insights management.
Broader inclusivity and deeper engagement
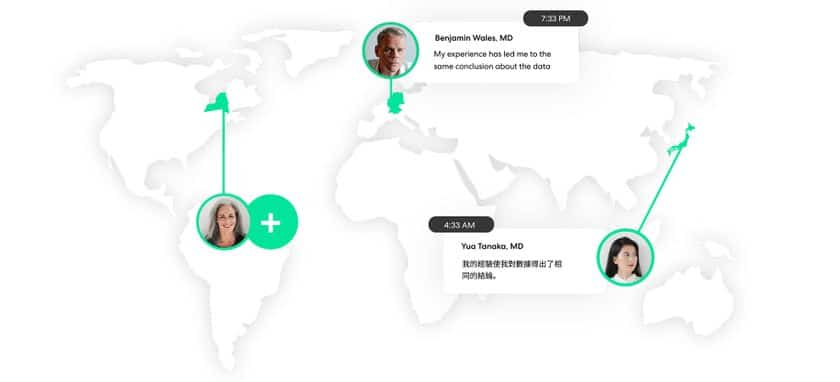
Virtual meetings offer the advantage of equal access for all interested attendees. With face-to-face engagements, individuals who couldn’t be present for some reason – schedule, cost, or other limitation – have to forfeit the experience, the knowledge they could have gained, and the insight they might have contributed. And research shows that for some audiences, a preference for virtual (or at least the option of virtual) will persist regardless of the reason – 84% of HCPs say they prefer virtual engagement to continue, preferring either all-virtual or a combination of virtual and in-person.
So what’s the benefit of taking a virtual-first approach to most collaboration? For one thing, conversations can go far beyond what’s possible in a time-limited environment. Follow-up questions can be asked. And people have the time to consider their answers and contribute more meaningfully – a distinct improvement on the “I agree” habit in face-to-face meetings.
In-person and virtual can co-exist and complement one another, and insights management technology makes the partnership even more successful. When multiple insight streams are managed in a single platform, the outcome of all those interactions is even more valuable. With a broader reach, deeper conversations, and more efficient processing of information, technology supports our increasingly multi-channel industry.
Improved resource efficiency
In the lead-up to in-person events like medical congresses, teams expend a lot of energy sussing out potential topics of discussions, identifying trends, and seeking out the KOLs and rising stars they’ll want to talk to. Turning these tasks over to technology like social media listening can improve this process and let team members focus on other critical work. Time saved in monitoring and analyzing large amounts of data can be better used to set those critical KOL meetings and refine topics for discussion.
Even when pharma and medical device teams plan smaller events like advisory boards or steering committee meetings, technology can ensure any time spent in-person is more effective. Whereas traditional follow-up questions may be shared over email or answered via phone, technology keeps all these insights in one place. Creating a stronger scientific narrative is easier when everyone has access to every observation, rather than just a slice of the whole conversation.
Don’t forget that virtual engagement offers efficiency gains for attendees, too. No requirement to miss work or leave family means no need to reschedule shifts, work ahead, or arrange childcare. Online advisory boards mean KOLs don’t spend several days traveling for a meeting that likely only spans a few hours. And let’s not ignore the fact that real-time virtual events like investigator meetings or congresses usually begin and end more or less on time, so attendees can plan around the virtual event agenda with minimal interruption to their regular daily schedules.
Sustainability and growth
Sure, virtual engagement does away with some amount of travel. But it’s more sustainable in other ways, too – it reduces workloads, makes better use of internal resources, and ultimately can improve outcomes for patients – all of which go hand-in-hand with many companies’ strategic objectives. The challenges of the last two years gave life science companies little time to make the switch from in-person first to virtual first, but with the benefit of time, increased know-how, and lessons learned, teams can begin to plan a more effective virtual-first insights management strategy – intentionally.
Quick takeaway: how do you know you need insights management? Check out the five signs you might be a good candidate, and download to send and share (no email address required).
[/vc_column_text][/vc_column][/vc_row][vc_row type=”full_width_background” full_screen_row_position=”middle” column_margin=”default” column_direction=”default” column_direction_tablet=”default” column_direction_phone=”default” bg_color=”#d6fff1″ scene_position=”center” text_color=”dark” text_align=”center” row_border_radius=”none” row_border_radius_applies=”bg” overlay_strength=”0.3″ gradient_direction=”left_to_right” shape_divider_position=”bottom” bg_image_animation=”none” shape_type=””][vc_column column_padding=”padding-10-percent” column_padding_tablet=”inherit” column_padding_phone=”inherit” column_padding_position=”all” column_element_spacing=”default” background_color=”#d6fff1″ background_color_opacity=”1″ background_hover_color_opacity=”1″ column_shadow=”none” column_border_radius=”none” column_link_target=”_self” gradient_direction=”left_to_right” overlay_strength=”0.3″ width=”1/1″ tablet_width_inherit=”default” tablet_text_alignment=”default” phone_text_alignment=”default” bg_image_animation=”none” border_type=”simple” column_border_width=”none” column_border_style=”solid”][vc_column_text]
More insights, direct to your inbox.
[/vc_column_text][nectar_btn size=”large” button_style=”see-through-3″ color_override=”#000000″ hover_color_override=”#04e499″ hover_text_color_override=”#ffffff” icon_family=”none” el_class=”black_button” button_id=”black_button” text=”Get our newsletter.” url=”https://resources.within3.com/newsletter”][/vc_column][/vc_row]